Words That Make Science Click
An Introduction to a Framework for Science Vocabulary Instruction
Making Vocabulary Work in the Science Classroom
Every semester, I focus on a specific goal to improve my science instruction. For Fall 2025, I decided to focus on vocabulary because I felt that the strategies I was using in my classroom were not as effective as I wanted them to be. For years, I had students study vocabulary through retrieval using flashcards. I now use a different retrieval system but if you are curious, you can check out my old flashcard system here. While this flashcard system was useful, I still felt something was missing. I knew I haven’t provided my students enough guidance to help them understand and use vocabulary in a meaningful way, which limited the results I hoped to see and interfered with my students’ ability to understand and develop explanations of scientific ideas. But I did not know how else to approach it.
Luckily, Sean Morrisey was able to help jumpstart my journey into vocabulary instruction (if you have not checked out his Word Mapping Project, please do so!). I saw a preview of his ResearchED vocabulary presentation on Twitter and decided to contact him. He was kind enough to schedule a Zoom meeting with me to discuss more. In this conversation, he mentioned that it is important for teachers to focus on Tier 2 vocabulary instruction because it bridges the gap between everyday language and complex academic language. After this discussion, I went through a rabbit hole that led me to the resources below:
Bringing Words to Life
A research-based guide that explains classroom strategies for teaching vocabulary to K-12 students. It explains how to choose which words to teach, how to introduce and reinforce word meanings, and how to connect vocabulary instruction to reading and writing.
To The Classroom Podcast
In this podcast episode, Jennifer Serravallo and Dr. Margaret McKeown (one of the authors of Bringing Words to Life book) discuss how to choose words for instruction and how to explicitly teach words so students can understand them deeply.
These resources were a great starting point and helped me deepen my understanding of how words support learning. It made me realize that in science, students don’t only need to understand subject-specific words that represent complex and abstract ideas, but must also learn the meaning of academic words that are used to describe them. Through this lens, I was able to see how much of a difference vocabulary instruction can make in a science classroom.
Let me provide an example.
Science Classroom Example
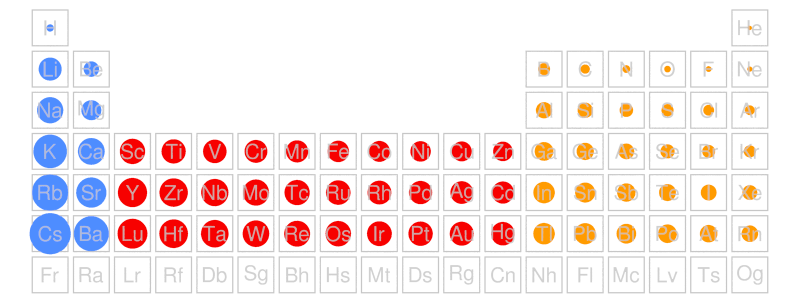
In chemistry, students are taught periodic trends. Periodic trends help students predict physical and chemical properties of elements based on their placement on the periodic table. For example, students are able to predict the relative size of an atom by looking at its placement on the periodic table:
As you move from left to right across a row (period), the size of the atom decreases.
As you move from top to bottom down a column (group), the size of the atom increases.
If students are asked which atom is bigger, sodium (Na) or rubidium (Rb), all they have to do is notice that sodium (Na) and rubidium (Rb) are in the same group and determine which one is lower in the group. Simple. Right?
No. It is not. My students struggle with this concept year after year. Eventually they get it but why does it take them so long to understand it? How could they not see it? It’s literally right there in the image!? It always drove me nuts.
And then one day, it clicked during a student interaction:
Student: “Miss, I still don’t understand.”
Me: “Find sodium."
Student: “Found it.”
Me: “Now find rubidium.”
Student: “Okay. Got it.”
Me: “Are they in the same group?”
Student: “Yes.”
Me: “We know that as we go down a group, from top to bottom, the size of the atom increases. So which element is at the bottom?”
Student: “Rubidium.”
Me: “So which one is bigger?”
Student: “But you said it increases as you go down a group? What does size have to do with the number of atoms?”
Me: “I’m sorry…can you clarify? I don’t understand what you are saying.”
Student: “You said increases. So more atoms. What does the size have to do with how many atoms there are?”
Let’s stop and analyze this interaction for a bit.
Here the student interpreted increases using its most common everyday schema: increase —> more of something. This is because the word increase is more often associated with quantity rather than magnitude in everyday experiences (and math classes). Take a look at the everyday examples below:
The number of students in the class increased this year.
The amount of money in the account increased.
The number of cars on the road increased during rush hour.
In each of these examples, increase refers to more of something rather than something becoming larger in size. This everyday use can easily lead students to interpret scientific statements like “size of atom increases” as meaning there are more atoms, rather than each atom is larger. As a result, when I explained to students, “As we go down a group, the size of the atom increases,” students interpreted this to mean that there are more atoms. This confused students because they were trying to see the nonexistent relationship between the size of the atom and the number of atoms.
This was a small discrepancy that I would have never noticed without this student interaction, but made such a huge impact on how students interpreted my original explanation during explicit instruction.
Since many students were struggling, I assumed that others had the same misconception. So I went back to the whiteboard to explain again but this time I explained the concept first and then clarified what I meant by the word increase:
“As we go down a group, from top to bottom, the size of the atom gets bigger. It goes from small to big as we see here.”
I wrote ‘small → big’ on the whiteboard that displayed the group from the periodic table.
“I’m going to ask you to find an element on your periodic table and point to it with one hand. Once you do that, raise your other hand. Find sodium.”
All hands went up.
“Great. Now do the same and find rubidium on the periodic table.”
Again, all hands went up.
“Take a few seconds to think about the next question. Do not say the answer out loud. Are these elements in the same group?”
I gave wait time.
“Hmmm…are these elements in the same group…Joel?”
“Yes.”
“That is correct. They are in the same group. Okay everyone…take a few seconds to think about the next question. Don’t say your answer out loud. Which element is lower in the group: sodium or rubidium?”
I gave wait time.
“On the count of three say the answer out loud as a class. One… two… three…”
“Rubidium!”
“Yes. Because rubidium is lower in the group, it is bigger than sodium as we can see in the image. Since atoms go from small to big as we move down a group, we use the academic word increase to describe this pattern. Repeat after me: increase.”
“Increase.”
“So the size of the atom INCREASES as we go down a group. What does this mean?”
I gave wait time.
“The size of the atom INCREASES as we go down a group. What does this mean? Hmmmm….Cristina.”
“It means that the atom gets bigger.”
“Yes. Okay, as a class, what does it mean? One…two…three.”
“Gets bigger.”
“What gets bigger? Hmmm..Karen.”
“The atom.”
“Yes. The atom gets bigger as we go down a group. And what academic word do we use to describe this pattern?”
I gave wait time.
“As a class on three…one…two…three.”
“Increase.”
“Very nice!”
I repeated this same structure to show that as we move across a period from left to right, the size of the atom decreases. We then examined examples and nonexamples using the same descriptors, followed by a check for understanding using mini whiteboards.
After this, the number of students who understood the concept increased (ha!) as evidenced by how they approached the problems on their miniwhiteboards. Students then moved on to independent practice.
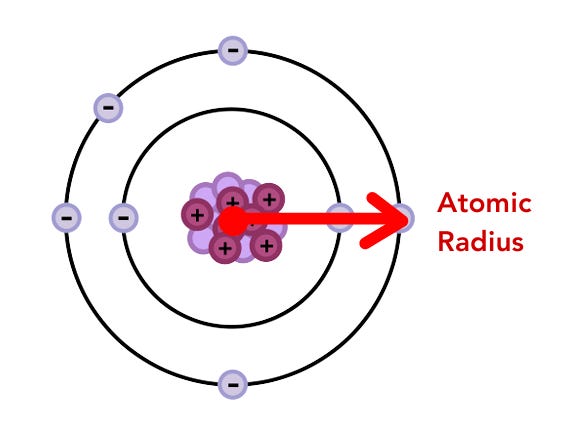
After independent practice, we reviewed the problems as a class for accuracy. I then introduced the word atomic radius.
“So far, we’ve been using the phrase size of the atom. In chemistry, scientists use a more precise term to measure this: atomic radius. Repeat after me. Atomic radius.”
“Atomic radius.”
“Good. Atomic radius is the measurement scientists use to describe how big an atom is. Just like a circle has a radius, atoms do too. The atomic radius tells us the distance from the nucleus to the outermost electron. Instead of scientists saying ‘the atom is big,’ they say ‘the atom has a large atomic radius.’”
Let’s take a look as to why I introduced the term atomic radius last.
Students had already interacted with the phrase “size of the atom” multiple times through examples and practice problems. When I introduced the term atomic radius, it simply became a label for an idea students already understood, so the definition made sense immediately. This made it easier for students to recall the definition during retrieval practice.
Words That Make Science Click Series
By being intentional on how I used vocabulary in my explanation, it improved student understanding. In the aforementioned example, I made modifications on the spot, so it was quite messy but over the course of the semester, I developed more structured systems that helped me teach vocabulary with greater intention and consistency.
I now want to share these strategies and systems in more detail in the hopes of receiving constructive feedback and offering other science teachers a place to start with evidence-informed vocabulary instruction. Even if you are not a science teacher, I believe many of these insights will be applicable to your classroom.
Because there is too much to share in a single blog post, I have turned this into a multi-part series that I call the Words That Make Science Click Series:




I love this series! Friendly advice: link Part I, II, and III from the bullets in the bottom of this post, and mirror those links in each piece, to make it easy to navigate across pieces.
Now off to find Pari I...
I love how a simple conversation with a student can sometimes unlock the key to explaining something better :)